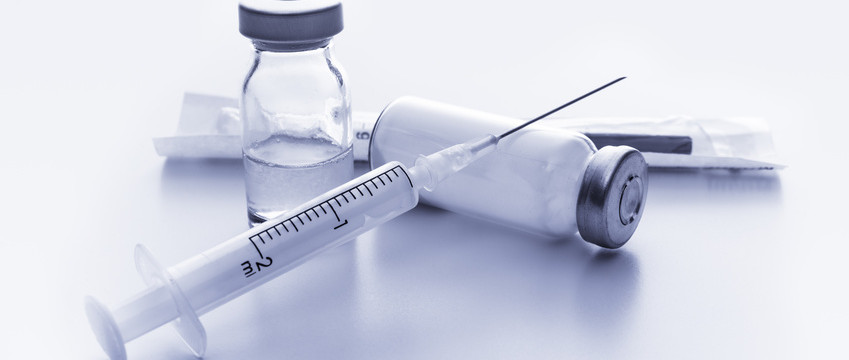
The approval has been received from US Food & Drug Administration (USFDA) for Aurobindo Pharma to manufacture Medroxyprogesterone Acetate Injectable Suspension USP with 150 mg/mL, 1 mL single dose Vial to be bioequivalent and therapeutically equivalent to the reference listed drug (RLD), Depo-Provera Injectable Suspension, of Pfizer Inc.
- Ashoka Buildcon JV Bags Rs 1,041-Crore BMC Flyover Project; Shares End 2.30% Higher
- WPIL Shares End 7.75% Higher on Order Win
- Biocon Launches Diabetes, Obesity Drug-Device in Netherlands; Shares Trade Flat
- Bharat Electronics Shares Trade Higher on Rs 776 Crore Orders
- Granules India Shares Slump Despite Securing EIR from USFDA
In the third quarter of next year, this product will be launched with an estimated market size of around US$ 62 million for the twelve months ending June 2022, according to IQVIA.
This is the 147th ANDA (including ten tentative approvals received) out of Eugia Pharma Speciality Group (EPSG) facilities, manufacturing both oral and sterile speciality products. Medroxyprogesterone Acetate Injection is used to prevent pregnancy for a female’s reproductive potential.
On the other hand, Aurobindo Pharma Limited (APL) ‘s shares were traded at Rs 559.60 close yesterday, which is compared to the previous close of Rs 551.80. The total number of share days was 51246 in over 2236 trades. The company’s stock hits a high of Rs 560.90 and a low of Rs 546.40 on an intraday basis. During the day, the net turnover was Rs 28488804.
 Live
Live